Uses of Calcium Chloride
When a no chemist thinks of salt, NaCl (sodium chloride) comes instantly to mind, but for a chemist, many other compounds also fit the bill, and CaCl2 is one of them. The CaCl2 compound name is calcium chloride. Like table salt, it’s a white crystal at room temperature, but it has properties that clearly set it apart. Calcium chloride can be obtained by refining soda ash, natural brine, or limestone. It dissolves readily in water and will actually turn to liquid if left long enough in a humid environment. Common uses include deicing roads, accelerating the set time of concrete, controlling humidity, and improving the taste of beer.
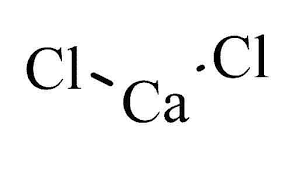
CaCl2 Uses on Roads and Highways
Like sodium chloride, calcium chloride lowers the melting point of ice, so one of its most common uses is for road deicing. It works at much lower temperatures — minus 20°F vs. 20°F for rock salt — because it actually releases heat in an exothermic reaction when it dissolves. It costs more per pound than rock salt, but you need less of it to do the same amount of deicing. Deicing products often contain both sodium chloride and calcium chloride.
Calcium chloride is hygroscopic, which means it attracts moisture from its environment. This quality makes it a good chemical to use for suppressing dust on gravel roads and other byways.
Calcium Chloride for Humidity Control
CaCl2 is more than just hygroscopic; it is deliquescent, which means it can absorb enough moisture to turn to liquid brine at room temperature. It’s the most common ingredient in moisture absorbers available at big box stores for controlling humidity in drawers and closets. You often find it combined with baking soda, which is a deodorizer.
Dehumidifying with calcium chloride is not without its hazards. CaCl2 is corrosive and promotes rust, so it should not be used around metals. Moreover, prolonged exposure can cause respiratory issues and lung damage. Silica gel is a safer desiccant and is often used instead of calcium chloride.

Calcium Chloride uses in the Food Industry
Calcium chloride is safe to consume and is a common food additive. It adds firmness to cheese, tofu, and canned fruits and vegetables, and it provides a way to enhance the salty taste of pickles without adding extra sodium. Brewers add it to the water they use to make beer to lower the pH and add alkalinity, which improves the taste.
Calcium chloride is a common ingredient in sports drinks. When it dissolves in water, it splits into positive calcium ions and negative chlorine ones, and because the calcium chloride formula includes two chloride ions, it creates a more powerful electrolyte than sodium chloride, which has only one. The ability of CaCl2 to completely dissociate into its component ions gives the compound a salty taste, and it could be a substitute for table salt were it not so hygroscopic. However, its propensity to absorb water causes intestinal problems and abdominal pain if you ingest it in large amounts.
Using Calcium Chloride in Construction
Because calcium chloride absorbs water so well, and it generates heat when doing so, cement workers add it to concrete to accelerate the cure rate at low temperatures. The addition of a 2 percent concentration creates the same cure rate at 50°F as plain concrete has at 70°F. Kits for testing the moisture content of concrete slabs typically contain calcium chloride in a container. Because CaCl2 absorbs off-gassing water vapor from the slab, the test consists of weighing the container, leaving it on the slab for a prescribed period, weighing it again, and recording the gain in weight.
Calcium Chloride for Pools


Uses of Calcium Chloride
The environment influences pool water–from leaves swirling down, to the swimmers, to airborne dust particles. All this affects the quality of the water, thus the need for constant cleaning and filtration. In addition, the water should have a proper balance of acidity, alkalinity, and calcium hardness. Calcium chloride maintains calcium hardness.
Uses of Calcium Chloride in Hard and Soft Water
Hard water is water oversaturated with calcium and magnesium. This excess calcium settles on the pool surfaces creating rough spots. It looks like a crystallized, whitish scale and feels like a rough spot. Soft water is low in calcium carbonate–it is undersaturated. Here the water becomes corrosive in its attempt to obtain the calcium. It pits pool surfaces and corrodes pipes and fixtures.
Correcting Pool Balance with Calcium Chloride
You can correct both hard and soft water conditions. If the pool water is undersaturated, then add calcium chloride to bring the level in balance. If the pool water is hard then you have to reduce the amount of calcium chloride in the water to balance it.
How to Use Calcium Chloride in Pools
The proper way to perform the addition of calcium chloride is to add a little at a time. Wait for it to dissolve, (about four hours) and then test the water. Add more only if needed. Thus, you do not oversaturate the water. There are three ways of reducing the amount of calcium chloride. You can dilute the pool water with fresh water, or add sequestering agents that keep the calcium in suspension. In extreme cases, you may have to drain the pool completely. The recommended hardness range is 200-400 ppm. Test weekly for the hardness level.
ARAX CHEMISTRY is a great manufacturer of Caustic Soda Flakes, Aluminum Sulfate, and Copper Sulfate which offers its High-quality products.

