Applications of caustic soda in industrial gas purification
Caustic soda is widely used in industrial gas purification processes due to its ability to remove impurities and contaminants from gas streams. Caustic soda is also known as sodium hydroxide (NaOH). In this article, we are going to describe a comprehensive explanation of the applications of caustic soda in industrial gas purification, stay with us.
Gas purification by Caustic soda
In gas purification processes, caustic soda or sodium hydroxide is used as a cleaning agent to remove impurities and pollutants from gases. caustic soda can effectively absorb and neutralize acid gases such as sulfur dioxide (SO2), chlorine (Cl2) and hydrogen chloride (HCl). In addition, it can trap particulate matter, volatile organic compounds (VOCs) and other contaminants in the gas stream.
Purification of hydrogen by caustic soda
caustic soda is used in the purification of hydrogen gas. Hydrogen gas produced from different sources, such as natural gas steam conversion or conversion to biomass gas, contains impurities such as carbon monoxide (CO), methane (CH4) and sulfur compounds. Caustic soda is used to remove these impurities through various chemical reactions including water-gas shift reaction, methanation and absorption of sulfur compounds.
Removal of acid gas by caustic soda
Caustic soda reacts with these acidic components and forms water-soluble salts. Acid gases are corrosive, toxic or undesirable for various reasons. Caustic soda is used to remove acid gases such as hydrogen sulfide (H2S) and carbon dioxide (CO2) from industrial gas streams. This reaction can be shown as follows:
H2S + 2NaOH -> Na2S + 2H2O
CO2 + 2NaOH -> Na2CO3 + H2O
The salts resulting from this reaction can be easily separated from the gas stream and thus purified.
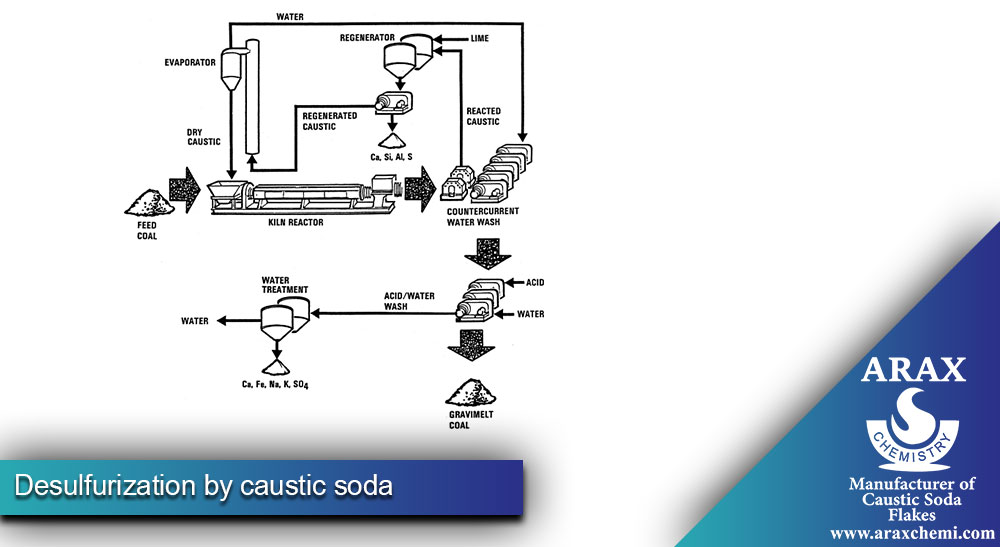
Desulfurization by caustic soda
Caustic soda plays a vital role in desulfurization processes. Caustic soda is used to remove sulfur compounds, especially hydrogen sulfide (H2S), which is commonly found in natural gas, biogas, and other industrial gases. These compounds should be removed due to their toxicity, corrosiveness and environmental effects. Caustic soda reacts with H2S to form sodium sulfide (Na2S), which can be further processed or safely disposed of.
Removal of mercury by caustic soda
Mercury is a highly toxic element found in some natural gas deposits and industrial emissions. Caustic soda is effective in removing mercury vapor from the flow of industrial gases. Caustic soda reacts with mercury to form stable complexes that can be easily removed from the gas stream using appropriate separation techniques.
Amine regeneration by caustic soda
Caustic soda helps remove contaminants from the spent amine solution and restores its efficiency for further gas treatment cycles. In gas purification using amines, such as gas sweetening or carbon absorption, soda mastic is used to regenerate the used amine solution. Amines selectively absorb certain gases such as CO2 or hydrogen sulfide from the gas stream.
pH adjustment with caustic soda
Sodium hydroxide is used to adjust pH in gas scrubbers and other gas purification systems. The pH of the gas stream is important for optimizing the efficiency of downstream processes or protecting equipment against corrosion. Caustic soda can be added to the gas stream to control and adjust acidity or alkalinity as needed.
The specific concentration and application of caustic soda varies depending on gas composition, process conditions, and regulatory requirements.
Water Gas Shift Reaction
Caustic soda is used in the water gas shift reaction, which is an important step in the conversion of carbon monoxide (CO) to hydrogen (H2). This reaction takes place at high temperatures and pressures and involves the interaction of CO with water vapor in the presence of a catalyst. Sodium hydroxide acts as a catalyst promoter and enhances CO conversion and facilitates hydrogen production.
Cleaning chlorine gas with caustic soda
Caustic soda is utilized in the scrubbing or absorption of chlorine gas (Cl2) from industrial gas streams. Chlorine gas is highly toxic and corrosive, and its presence in gas streams can be detrimental to downstream processes or equipment. Caustic soda reacts with chlorine gas, forming sodium hypochlorite (NaOCl) and sodium chloride (NaCl), which are less hazardous and easier to handle.
Cl2 + 2NaOH -> NaOCl + NaCl + H2O
The formed sodium hypochlorite can be further processed or safely disposed of.
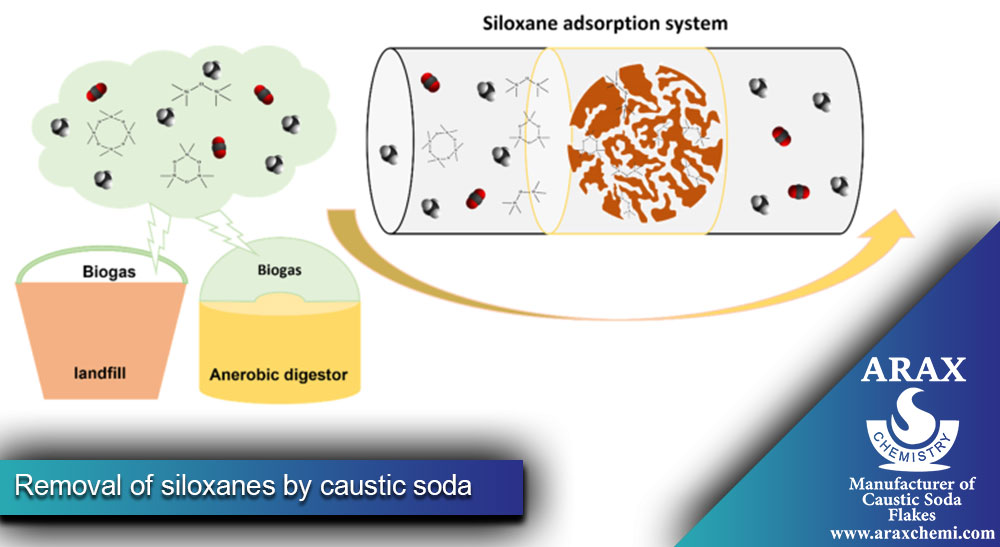
Removal of siloxanes by caustic soda
Siloxanes are silicon-oxygen compounds commonly found in biogas from organic waste. These compounds can cause serious damage to equipment such as gas engines or turbines when ignited. Caustic soda is used to remove siloxanes from biogas by a process called leaching or absorption. The caustic soda solution traps the siloxanes and prevents them from entering downstream equipment, minimizing the risk of damage.
Regeneration of Spent Caustic Solution
In certain gas purification processes, caustic soda solution is used as a washing or absorption medium to remove impurities. Over time, the caustic soda solution becomes saturated with the absorbed contamination.
Regeneration of absorbents by caustic soda
Caustic soda is used in the regeneration of adsorbents in gas purification processes. Adsorbents such as activated carbon or molecular sieves are often used to trap and remove impurities from the gas stream. When these adsorbents become saturated, they can be regenerated by washing with sodium bicarbonate solution, which helps to release and remove the adsorbed impurities, thus restoring the capacity of the adsorbent for further use.
Removal of volatile organic compounds (VOCs) by caustic soda
Volatile organic compounds are organic chemicals that can be present in the gas stream from various industrial processes. Caustic soda is used to clean or absorb VOCs such as benzene, toluene, and xylene. It reacts with these organic compounds and causes their dissolution or the formation of water-soluble salts. This helps remove VOCs from the gas stream and reduce air pollution.
Other uses of caustic soda in gas purification
Other uses of caustic soda in gas purification include the following.
- Oxygen removal: Oxygen can be an undesirable component in certain gas streams, such as those used in certain chemical reactions or in the production of electronic components. Caustic soda is used to remove oxygen through chemical reaction or absorption. Caustic soda reacts with oxygen to form sodium peroxide or sodium hypochlorite, which can be further processed or removed from the gas stream.
- Regeneration of catalysts: In specific gas purification processes, catalysts are used to facilitate specific reactions or increase the removal of impurities. Over time, these catalysts can become inactive or contaminated. Caustic soda is used in the regeneration of these catalysts using a caustic soda solution. This combination helps to remove accumulated impurities, restore catalyst activity and increase their lifespan.
- Removal of Particulate Matter: Caustic soda can be utilized in gas purification processes to remove particulate matter or solid particles suspended in the gas stream. It can be used as a wet scrubbing agent or as part of a filtration system to trap and remove these particles, ensuring cleaner and purer gas output.
- Biogas Upgrading: Caustic soda is commonly used in the upgrading or purification of biogas to produce biomethane, a renewable and high-quality substitute for natural gas. It helps in removing impurities such as carbon dioxide, hydrogen sulfide, siloxanes, and moisture, ensuring that the biomethane meets the required quality standards for injection into the natural gas grid or use as a transportation fuel.
Conclusion
In this article, we learned about some additional uses of caustic soda in industrial gas purification. The specific application and concentration of caustic soda may vary depending on gas composition, process requirements and industry standards. When using caustic soda in gas purification operations, it is necessary to follow appropriate safety protocols and consult with experts or refer to relevant guidelines. In the end, you can contact our experts for advice and purchase.